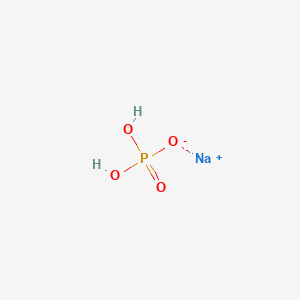
NaOH+H3PO4=NaH2PO4+H2O Balanced Equation||Sodium hydroxide+Phosphoric acid Balanced Equation - YouTube

Jual nah2po4 h20 sodium dihydrogen phosphate monohydrate merck 1.06346 - Kota Surabaya - mutiara bersaudara | Tokopedia
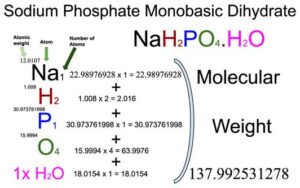
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes

Phase Equilibrium for the Ternary System KH2PO4 + NaH2PO4 + H2O at 303.15 K | Journal of Chemical & Engineering Data
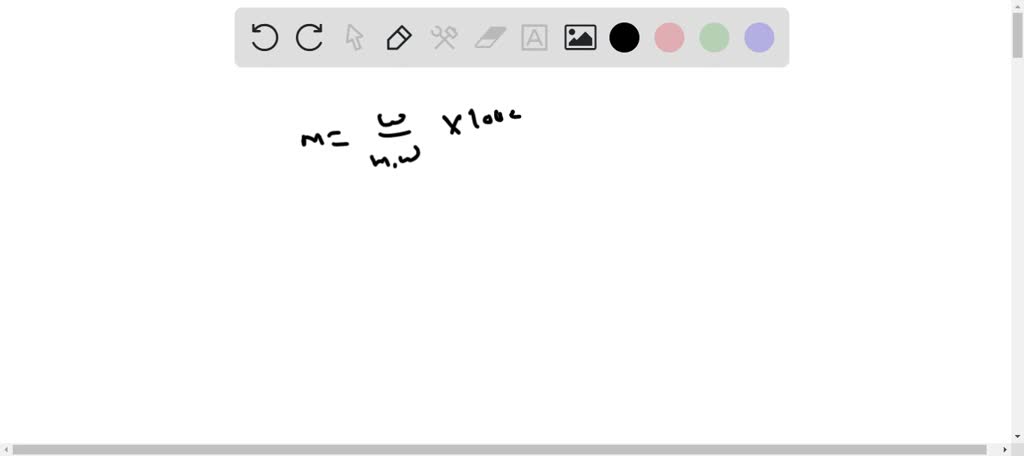
SOLVED: Weigh out 10.4 g sodium phosphate (NaH2PO4.H2O, MW = 138 g/mol). Dissolve the NaH2PO4.H2O in 450 mL water. Bring pH of solution to 7.4 with 10 M NaOH (need about 2

1 Bán Hóa chất Sodium dihydrogen phosphate monohydrat, reagent grade - NaH2PO4.H2O - SO0331 - Scharlau giá rẻ ở hcm

Jual sodium dihydrogen phosphate monohydrate merck 1kg / NaH2PO4. H2O - Kota Depok - LERES MART | Tokopedia
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%20500G.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs






![ACID-BASE BALANCE pH is a measure of H + pH = - log [H +] Importance: - ppt download ACID-BASE BALANCE pH is a measure of H + pH = - log [H +] Importance: - ppt download](https://slideplayer.com/slide/13040484/79/images/9/PHOSPHATE+BUFFERS+Important+in+kidney+Buffer+pair+%3D+Na2HPO4%2FNaH2PO4.jpg)