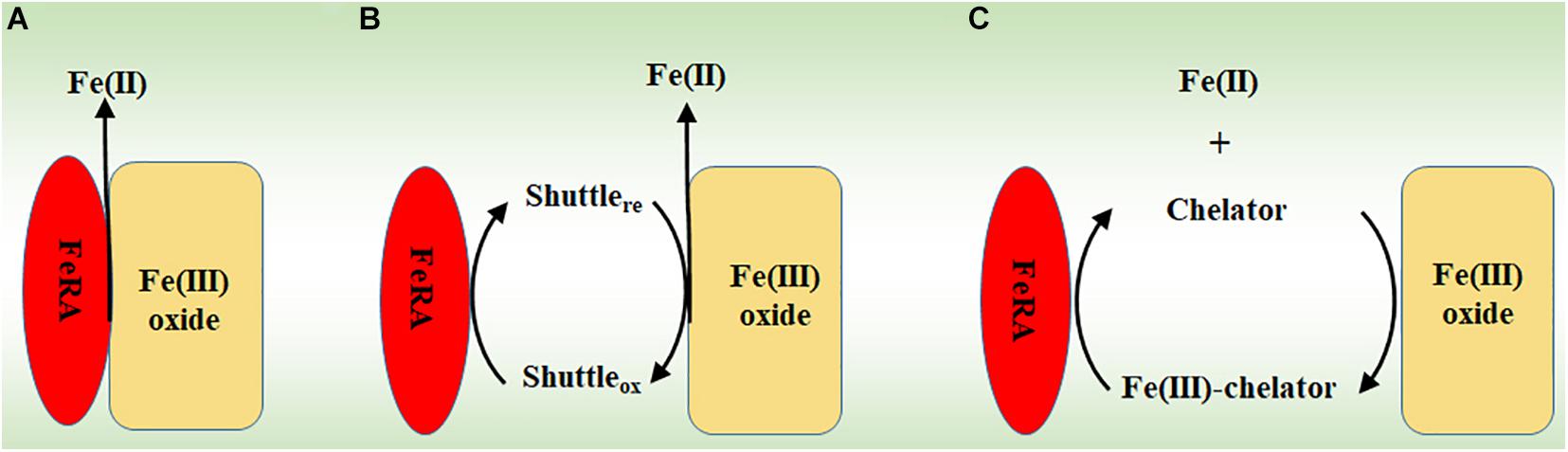
Frontiers | The Proposed Molecular Mechanisms Used by Archaea for Fe(III) Reduction and Fe(II) Oxidation
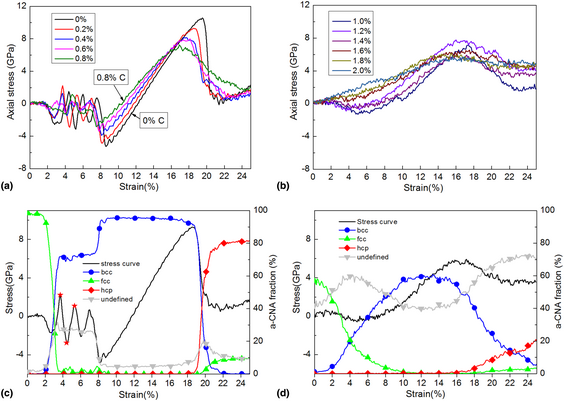
The strain-induced martensitic phase transformation of Fe–C alloys considering C addition: A molecular dynamics study | Journal of Materials Research | Cambridge Core
Iron is produced by the reduction of iron (III) oxide using carbon monoxide. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g). How much Fe is produced from 1 kg of Fe2O3? - Quora

Show the full molecular orbital diagram for \begin{bmatrix} Fe(OH_2)_6\end{bmatrix}^{2+}. Indicate all metal atomic orbitals, ligand LGOs, bonding MO's, nonbonding MO's, antibonding MO's, symmetry labels and \Delta_0. | Homework.Study.com
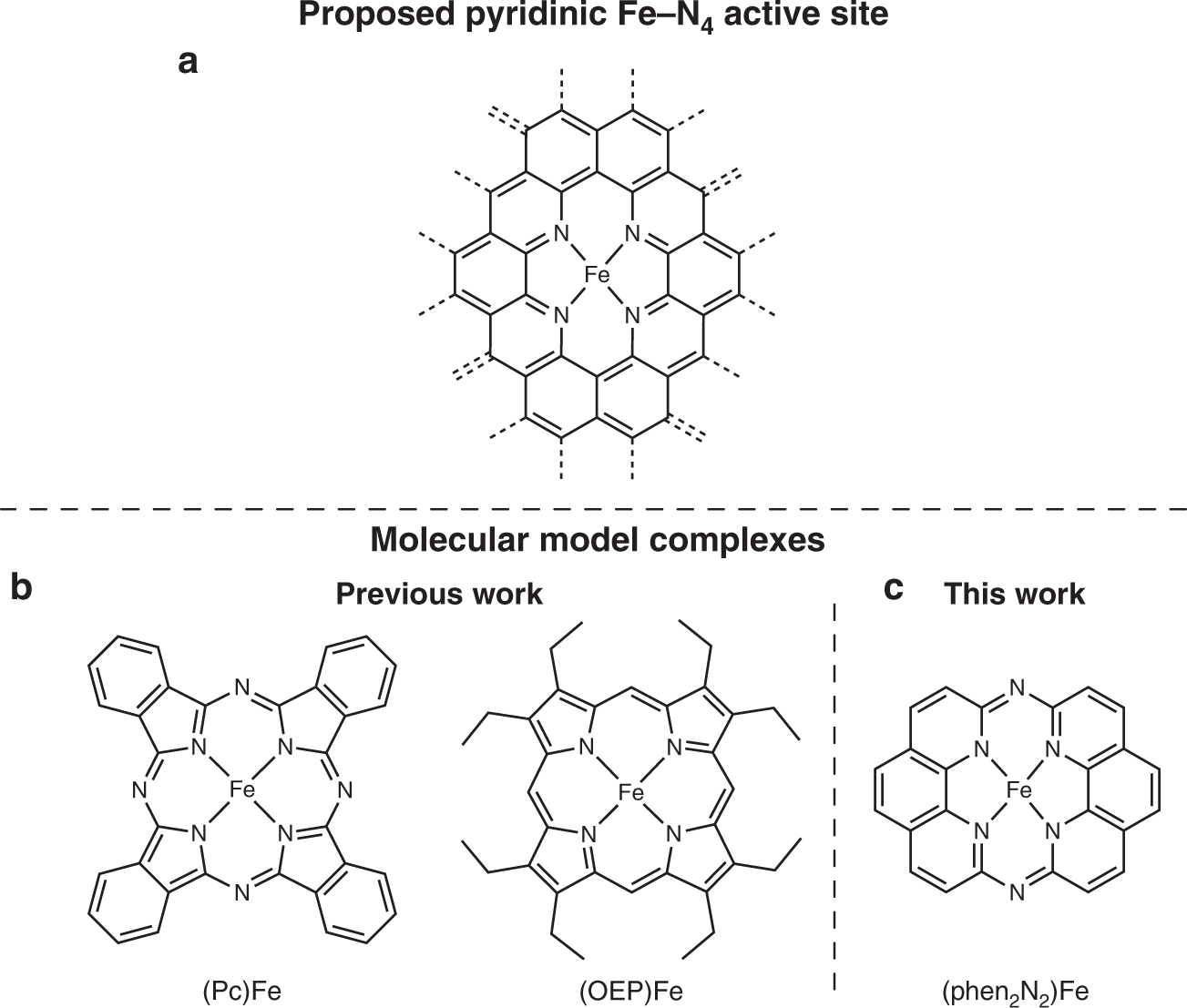
A pyridinic Fe-N4 macrocycle models the active sites in Fe/N-doped carbon electrocatalysts | Nature Communications
![Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation](https://pubs.acs.org/cms/10.1021/ct501143c/asset/images/medium/ct-2014-01143c_0017.gif)
Ground and Excited States of the [Fe(H2O)6]2+ and [Fe(H2O)6]3+ Clusters: Insight into the Electronic Structure of the [Fe(H2O)6]2+–[Fe(H2O)6]3+ Complex | Journal of Chemical Theory and Computation

Characteristics of Fe and Mn bearing precipitates generated by Fe(II) and Mn(II) co-oxidation with O2, MnO4 and HOCl in the presence of groundwater ions - ScienceDirect











