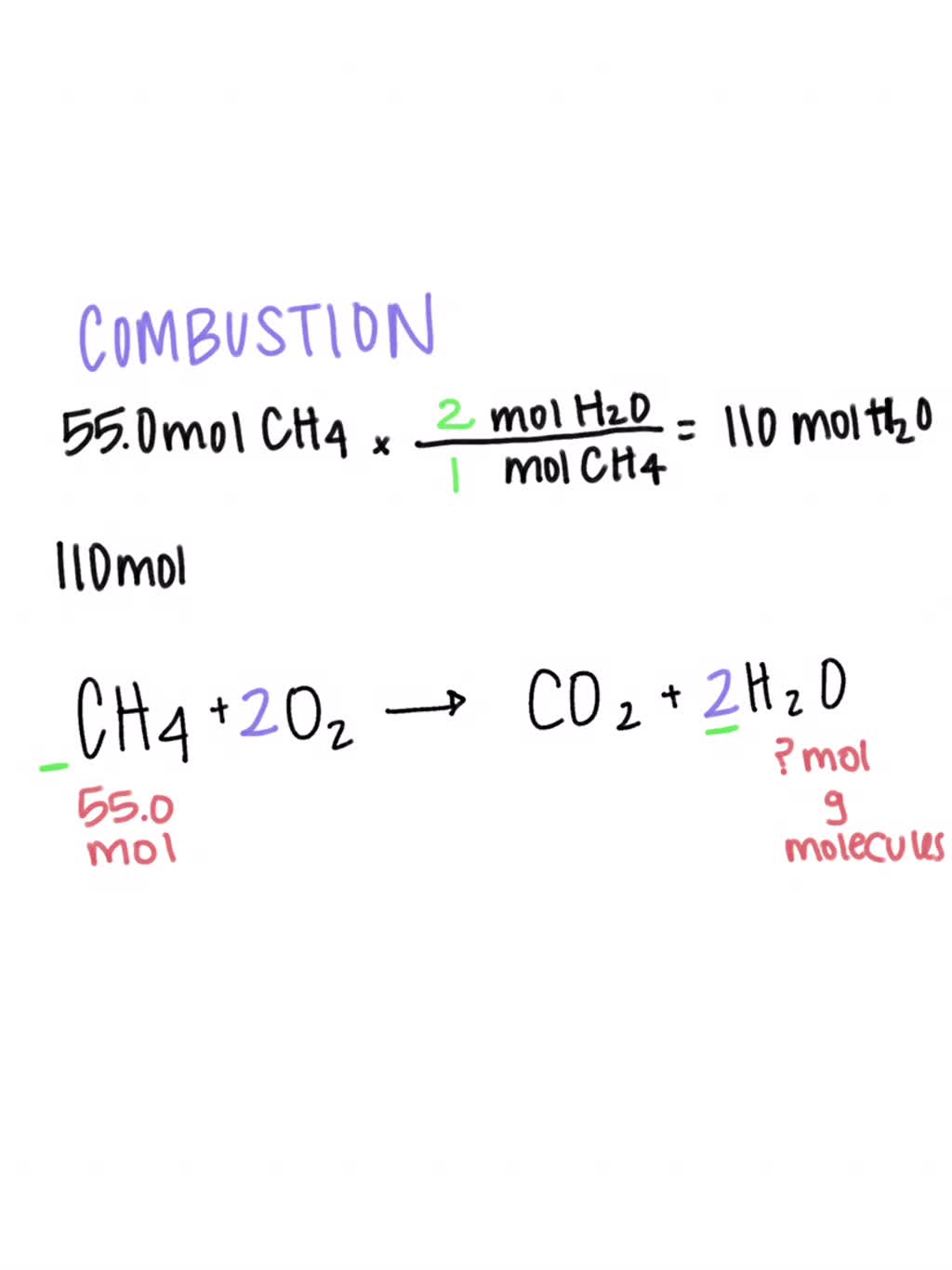
AMMIE ‼️ on Twitter: "HELP CHEM NERDS HOW DO I BALANCE C5H9O+O2=CO2+H2O becaUSE IM.. positive this is NOT right https://t.co/MxfFjIRsRn" / Twitter

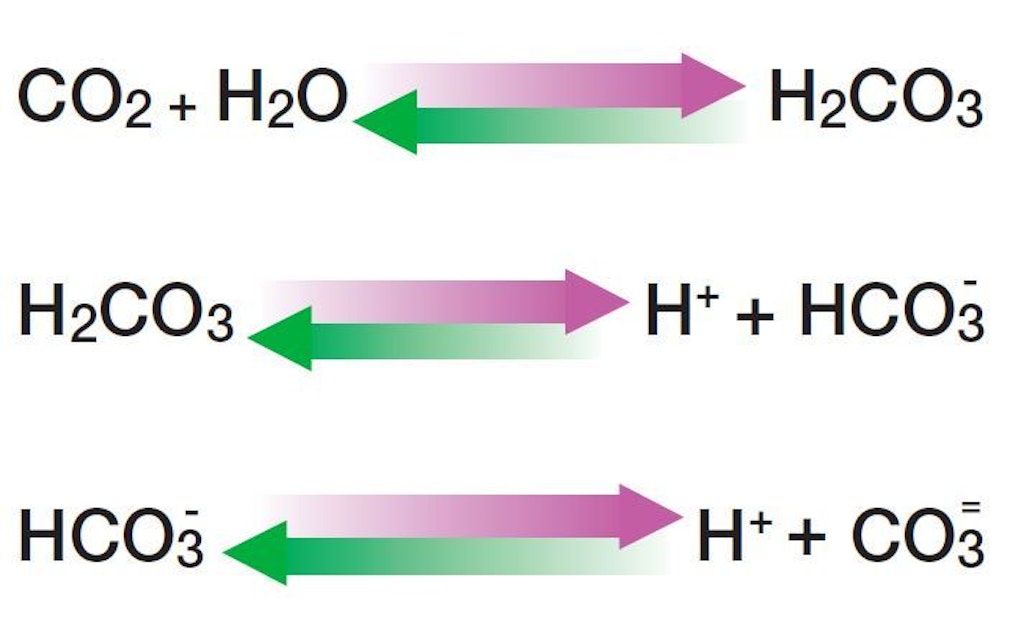
CO2+H2O=H2CO3 balance the chemical equation @mydocumentary838. co2+h2o=h2co3 balance the equation. - YouTube

The promoting effects of CO2 and H2O on selective hydrogenations in CO2/H2O biphasic system - ScienceDirect
![Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D](https://dwes9vv9u0550.cloudfront.net/images/4940114/ca6cb326-72a5-4e75-8a33-8b87ccbb46e4.jpg)
Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D
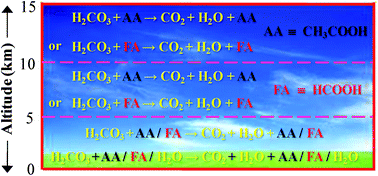
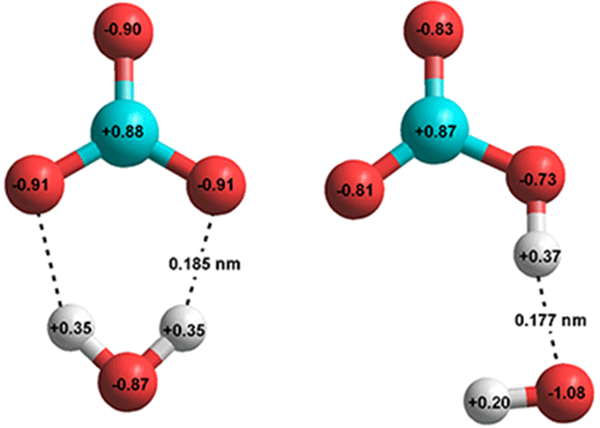
H2CO3 → CO2 + H2O decomposition in the presence of H2O, HCOOH, CH3COOH, H2SO4 and HO2 radical: instability of the gas-phase H2CO3 molecule in the troposphere and lower stratosphere - RSC Advances (
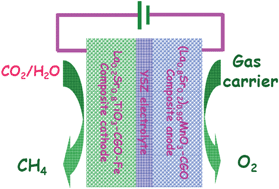
Direct synthesis of methane from CO2/H2O in an oxygen-ion conducting solid oxide electrolyser - Energy & Environmental Science (RSC Publishing)