Main-chain engineering of polymer photocatalysts with hydrophilic non-conjugated segments for visible-light-driven hydrogen evolution | Nature Communications
For a reaction, N2(g)+3H2(g) →2NH3 (g), the rate of formation of NH_3 was found to be 2 x 10^ 2 mol/dm3/s. The rate of consumption of N2 will b

SOLVED: Practice Problems 15. Determine the mass in grams of each of the following 3.57 mol Al b: 42.6 mol Si 16. Challenge Convert each given quantity in scientific notation to mass

Oxidation of secondary alcohols using 10 mol% lanthanum triflate as... | Download Scientific Diagram

Given the following UNBALANCED equation, determine how many moles of potassium chlorate are needed to produce 10.0 mol of oxygen gas. 2KClO 3 (s) → 2KCl. - ppt download

Nickel-Catalyzed Stereoselective Glycosylation with C(2)-N-Substituted Benzylidene d-Glucosamine and Galactosamine Trichloroacetimidates for the Formation of 1,2-cis-2-Amino Glycosides. Applications to the Synthesis of Heparin Disaccharides, GPI Anchor ...
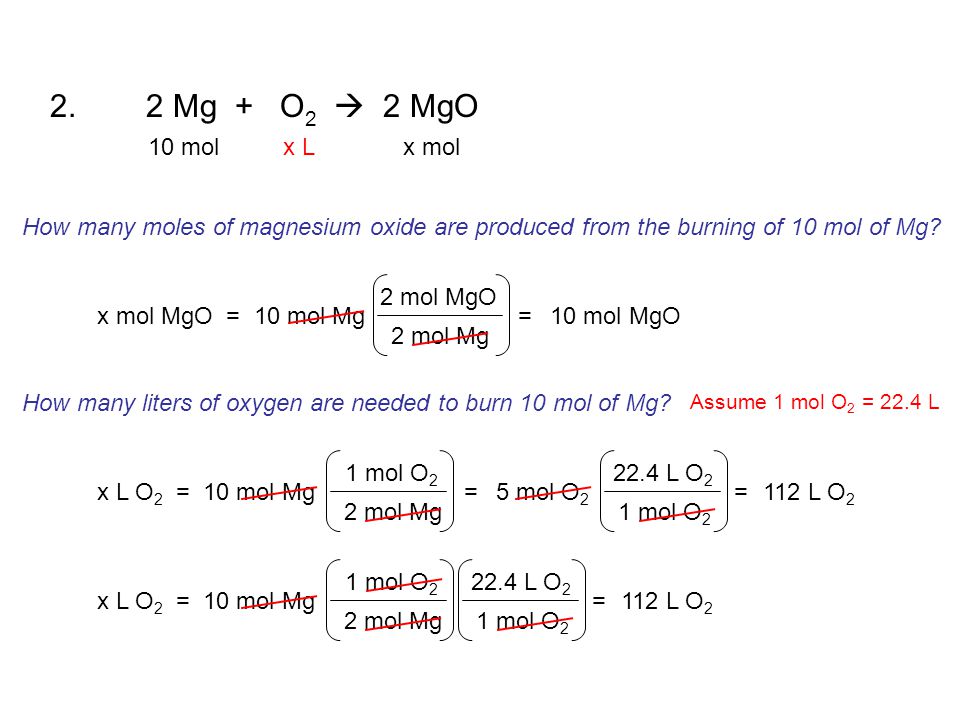
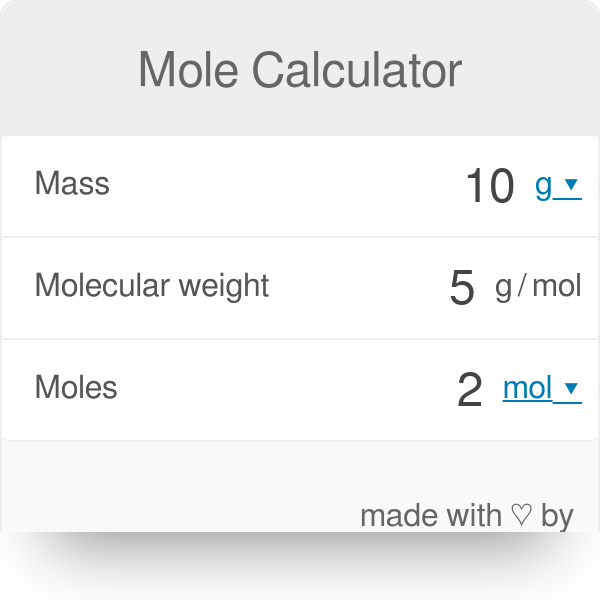
Mole Calculations. ? Visualizing a Chemical Reaction Na + Cl 2 NaCl ___ mole Cl 2 ___ mole NaCl___ mole Na ppt download