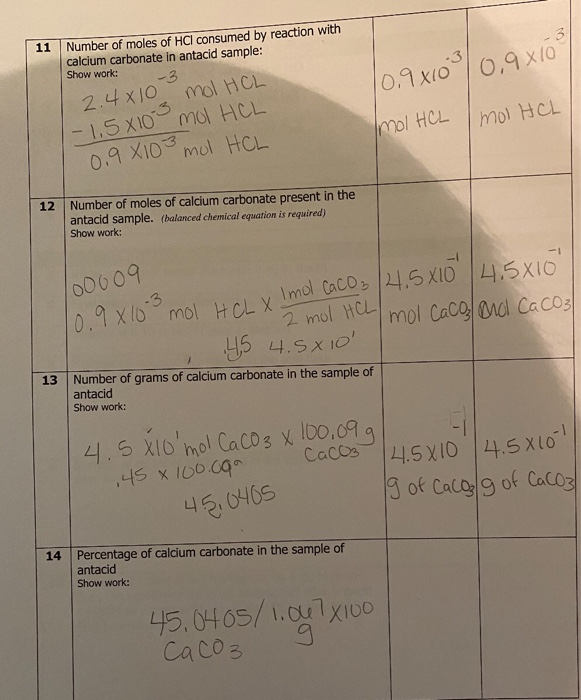
Effect of acid concentration on the oxidation of R·HCl. Acidity of the... | Download Scientific Diagram

SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole
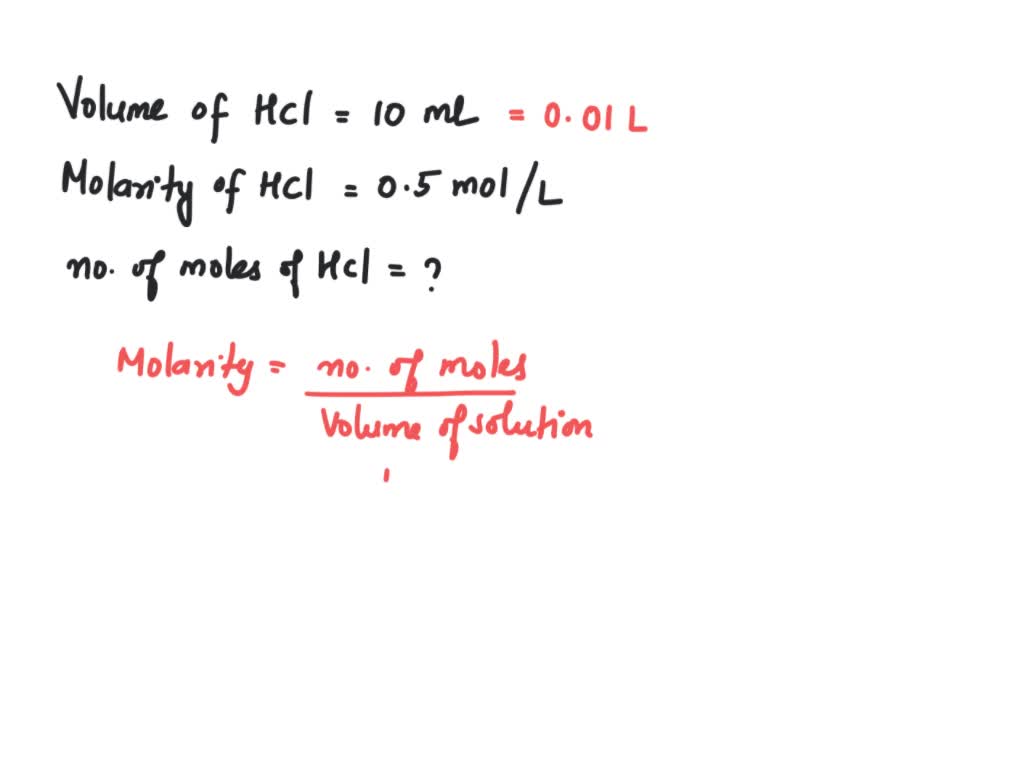
SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole
✓ Solved: Calculate the pH after 0.020 mole of HCl is added to 1.00 L of each of the four solutions in...
at 25 degree celsius the molar conductance at infinite dilution for HCl solution is 4.25*10 2 ohm m2 mol , while its specific conductanceis 382.5 ohm m . if degree of dissociation is 90
Assuming that: Part I (molarity of HCl by standardization): Part II (determination of sodium carbonate in unknown) Solution: Mas


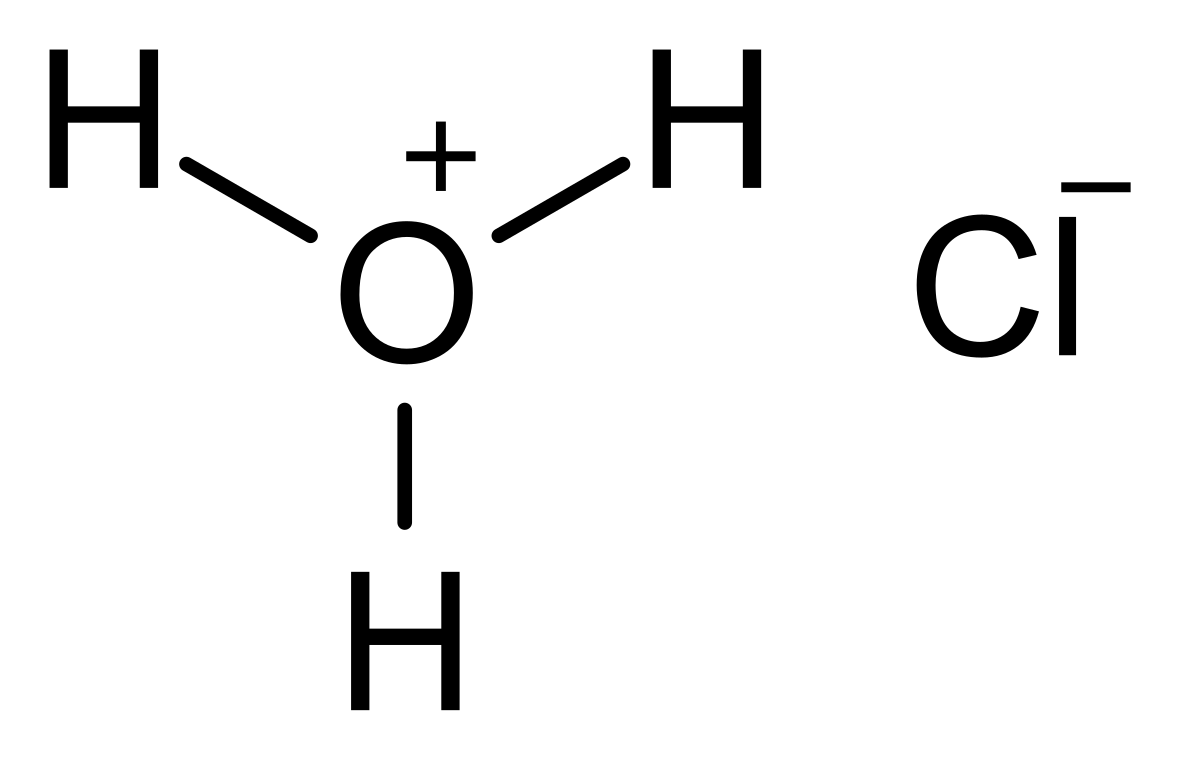
![ANSWERED] A flask contains 0.015 mol of HCl in 10.... - Inorganic Chemistry ANSWERED] A flask contains 0.015 mol of HCl in 10.... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/55747522-1659637855.5533836.jpeg)